 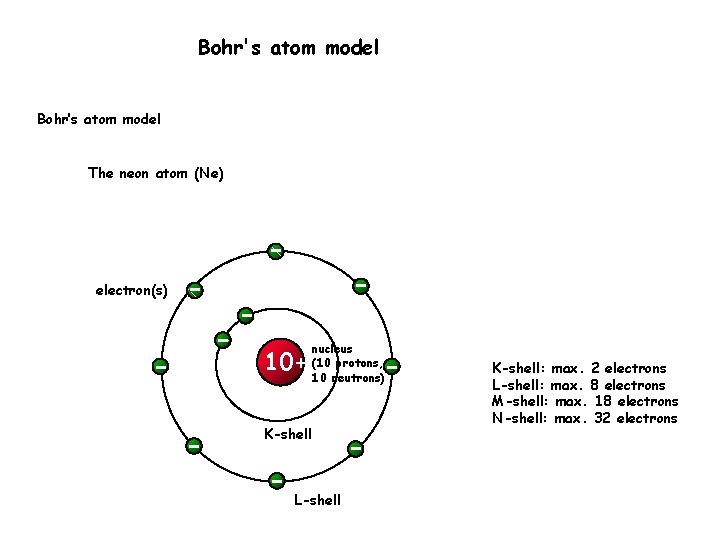
However, excited electrons will very quickly undergo spontaneous emission of a photon to return to a lower energy level. The only exception is when an electron has been excited. Each shell must be filled before the next starts to fill. The electron configuration of ten elements, Wikimedia Commons CC-BY-4.0Īs electrons are added to an atom, they will fill the lowest unfulfilled energy levels available. The diagram below shows the electron configuration of ten elements on the periodic table, assuming no electron has been excited to a higher state. The first shell can only hold two electrons, while shells 2 and 3 can contain 8 electrons each. 
There is a limit to how many electrons can physically exist in any shell at a given point in time. 6 eV, giving the electron ionization energy of 13. Consider a hydrogen atom with a ground state of - 13. This energy is defined as ionization energy or ionization state. The negative symbol denotes that the electron must be given energy to be ejected from the atom entirely. It’s important to note that the energy of an electron in an atom is always negative. The lowest level (E1) is the ground state, while all higher energy levels are excited states. They cannot exist between energy levels unless they are passing from one level to another. In Bohr’s model, electrons orbit the nucleus at discrete energy levels called shells. As an atom can only absorb and emit certain colors of light, we know that something within the structure of the atom must have specific energy levels.Īn electron absorbs and emits photons to move energy levels, Wikimedia Commons CC BY 3.0 Redder light has lower energies, while bluer light has higher energies. 
The energy of an EM wave determines its color. Remember that light is pure energy in the form of an electromagnetic wave or photon. The proton and the neutron (the subatomic particles that make up the nucleus) were discovered later, in 19, respectively. He proposed that the atom actually consisted of a small, compact, and positively charged nucleus surrounded by a cloud of electrons, called the Rutherford model. It took several years for Rutherford to correctly interpret the results of his experiment. However, a small number of the particles were deflected, which can be explained by Rutherford's Model, Wikimedia Commons CC-BY-SA-3.0 In the experiment with alpha particles, the expectation was for the alpha particles to go through without any deviation, in accordance with Thomson's model. However, a very small number of the alpha particles were deflected, sometimes being reflected completely. He expected the alpha particles to pass through the gold with no deflection as the positively charged "pudding" should be evenly spread out. Alpha particles are a form of radiation with a large positive charge. In 1905, Thomson’s student Ernst Rutherford tested the plum pudding model by directing a beam of alpha particles at a strip of gold foil. Thomson proposed that negatively charged "plums" (electrons) were surrounded by a positively charged "pudding," as an atom must contain some positive charge to cancel out the negative charge of the electrons. The model attempted to explain how an atom had no net electric charge, yet individual electrons possessed negative charges. After much deliberation and research, he proposed the plum pudding model or the Thomson Model. However, when the electron was discovered in 1897 by J. In 1803 John Dalton theorized that the atom was indivisible and could not be broken down into anything smaller. 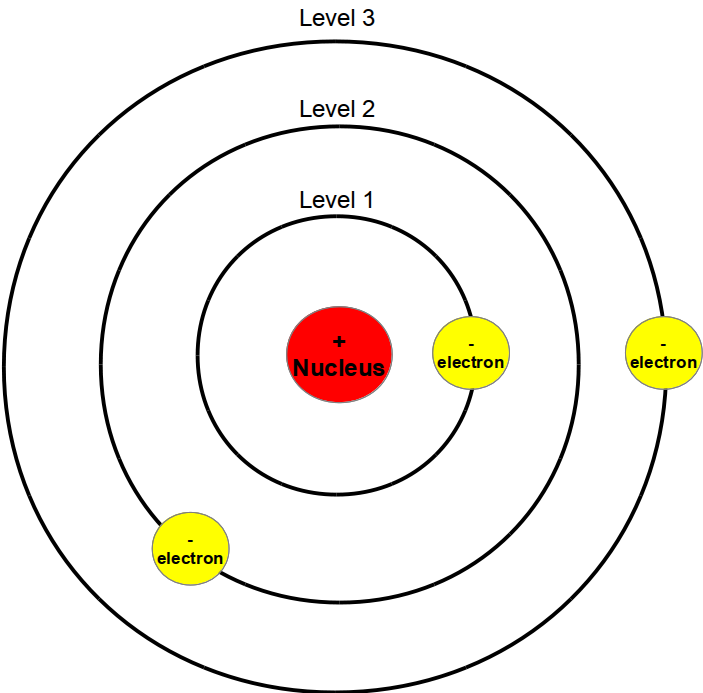
Plum Pudding Modelīefore the 20th century, we had no idea what the subatomic structure of the atom was like. Our understanding of the atom went through several distinct models throughout the last two centuries, improving with accuracy as new evidence was obtained and more fundamental physical principles were revealed. Development leading to the Bohr Model of the Atom Electrons can move between these shells by absorbing or emitting photons with specific energies. In the Bohr model of the atom, electrons can only exist in clearly defined levels called shells, which have a set size and energy, They 'orbit' around a positively-charged nucleus. In the Bohr model of the atom, electrons orbit around a positive nucleus. We have even learned that atoms are made up of even smaller subatomic particles and how they are structured within the atom. In the past 200 years, incredible advances have been made in the realm of atomic physics. You could line up over 300,000 Carbon atoms and they could still be hidden behind a single strand of human hair. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
